|
At the time it was known that DNA polymerase could copy DNA, and that it only went one way up the double helix, copying the so-called leading strand. Their key discovery came in the late 1960s. They decided to focus their collective scientific power on unravelling the mystery of DNA copying, or replication, investigating the intricate details of the process in frog and sea urchin eggs, moving between the US and Japan over the years. Besides research, their main hobby was heading out to the local noodle shop to watch Sumo wrestling on TV, as they had no set of their own. The roof of their lab leaked, and they often had to buy supplies out of their own pockets. But as long as Reiji had a position, she did too. It was a good move on Tsuneko’s part: at the time, it was very difficult for women to find jobs in science, apart from teaching, or even be recognised as researchers in their own right. The Okazakis got married later that year and decided to set up a lab as well as a home together, still at Nagoya University. So Tsuneko went to the local university to study biology, graduating with a PhD and a husband-to-be in 1956. It turns out there isn’t just one Okazaki – there’s two: Tsuneko and her husband Reiji.īorn Tsuneko Hara in 1933, in Nagoya, Japan, she was one of the first generation of Japanese women to take advantage of the country’s new post-war constitution, which allowed women to attend university alongside men. Well, I was not only wrong, but I was also suffering from internalised patriarchy. So, who was Okazaki? I always assumed that he was another of those guys from the classic era of molecular biology in the 1960s. (Oh, and if all of this sounds completely baffling, then check out this handy animation) This creates short stretches of newly-copied DNA – Okazaki fragments - with gaps in between, that have to be patched up in order to create a perfect new helix. For one side this is no problem - the polymerase can just run along and make new DNA in an unbroken chain.īut the other side has to do some molecular gymnastics, twisting around in a loop so it can be fed into the polymerase machinery from the right direction, then popping out, twisting again and feeding in a new stretch, a bit further along. So the two sides of the ladder have to be copied in different ways. For presumably boring reasons to do with evolution of protein structures, DNA polymerase, the enzyme that copies DNA, can only run in one direction. When DNA is copied, these two strands are pulled apart and complementary strands synthesised to match. If it helps, imagine two long chains of stacked pint glasses laid side by side, one stack all facing in one direction and the other running the opposite way. Each of the two side struts runs in an opposite direction, up and down (or ‘5 prime to 3 prime and 3 prime to 5 prime’ as biologists say), according to the directional stacking of the DNA letters, or bases, that make up the molecule. FEN 1 and RNase H1 remove the RNA from the Okazaki fragments and DNA ligase I joins the DNA.I first learned about Okazaki fragments in my undergraduate biochemistry class, when we learned how the DNA in every cell is copied so that it can divide.ĭNA is a double helix – a twisted ladder. PCNA and RFC function as a clamp and a clamp loader. 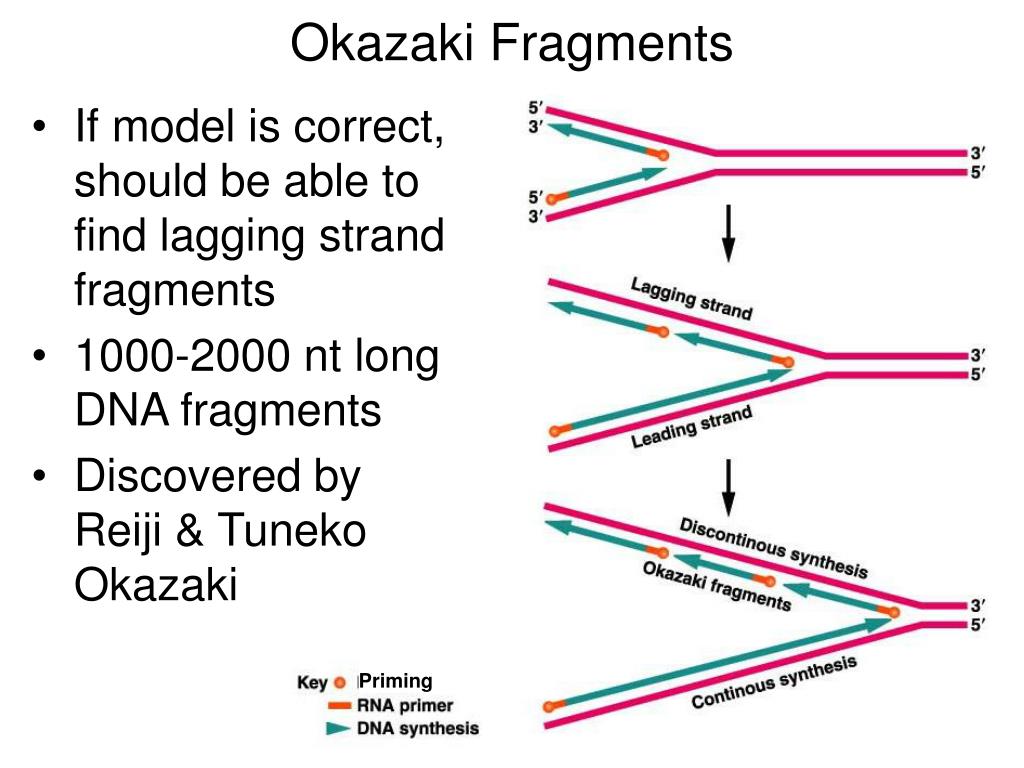
DNA primase forms a permanent complex with DNA polymerase alpha. In eukaryotes, three DNA polymerases (alpha, delta, and epsilon) have been identified. Normally, during replication of the lagging-strand DNA template, an RNA primer is removed either by an RNase H or by the 5 to 3 exonuclease activity of DNA pol I, and the DNA ligase joins the Okazaki fragments. The DNA primase (DnaG) is needed to form RNA primers. 
In prokaryotes, the leading strand replication apparatus consists of a DNA polymerase (pol III core), a sliding clamp (beta), and a clamp loader (gamma delta complex). On the leading strand, replication occurs continuously in a 5 to 3 direction, whereas on the lagging strand, DNA replication occurs discontinuously by synthesis and joining of short Okazaki fragments. At the DNA replication fork, a DNA helicase (DnaB or MCM complex) precedes the DNA synthetic machinery and unwinds the duplex parental DNA in cooperation with the SSB or RPA. Generally, DNA replication follows a multistep enzymatic pathway. A complex network of interacting proteins and enzymes is required for DNA replication.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
